Unlocking New Possibilities in
Cancer Treatment with MTL-004
In the realm of oncology, traditional cytotoxic drugs that damage DNA continue to be widely used, despite significant adverse side effects and patient concerns. This market remains robust, valued at over $26 billion annually.
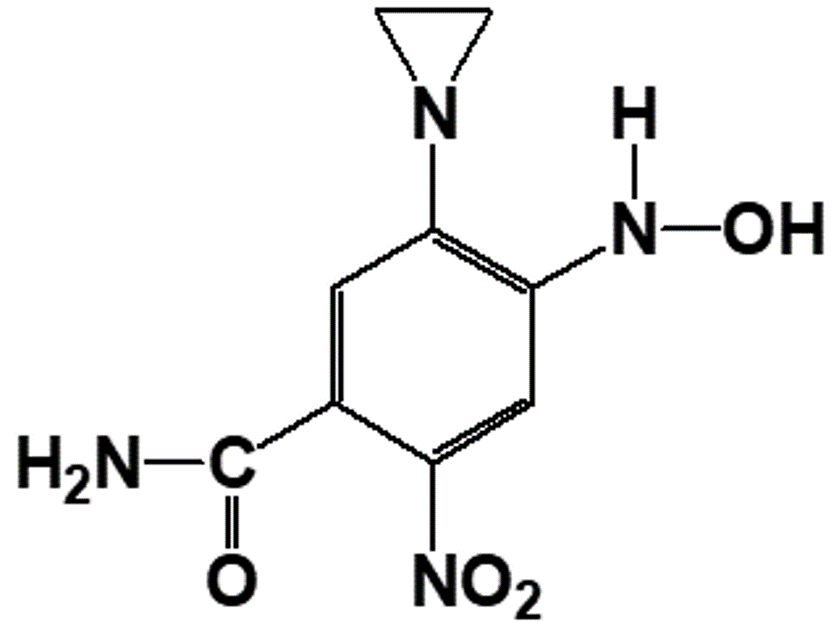
What is MTL-004?
MTL-004 is a proprietary small molecule drug candidate designed for the safe and effective treatment of solid tumours and prophylactic use at the margins of post-surgical resection sites. It induces localised apoptosis without the common side effects of less specific traditional cytotoxic therapies, ensuring its potent effects are confined only to targeted treatment areas.
Its unique formulation allows for enhanced therapeutic outcomes while prioritising patient safety, making it a promising option for precision treatments.
Key Highlights of MTL-004
Development and Market Potential
Regulatory Progress
The MHRA has reviewed MTL-004’s CMC, non-clinical safety, and putative efficacy data, providing scientific advice that enables Gordian to finalise MTL-004 for clinical evaluation for the treatment of Glioblastoma (GBM).
Fast-Track Approval
We are currently preparing orphan drug applications in both the EU and USA for the use of MTL-004 following tumour resection in GBM. These applications will offer ‘fast track’ routes to approval, supported by a UK clinical study slated for 2025.
Comprehensive Protection
MTL-004 is fortified by patents, Orphan Drug Designation, and Data Exclusivity, ensuring strong commercial protection as we advance.
Expanding Horizons
In addition to GBM, we have initiated work to evaluate MTL-004 as a post-resection treatment for Invasive Bladder Cancer (IBC) and have identified other accessible tumour sites for potential treatment with MTL-004.
Market Potential
The clinical use of MTL-004 holds the promise of a billion-pound market opportunity globally, paving the way for innovative cancer treatments that prioritise patient safety and efficacy.
The Need & Opportunity
Chemotherapy Remains in Widespread Use due to Clinical and Cost-Effectiveness ….. but has Significant Limitations
DID YOU KNOW?
• 1 / 3rd of patients fail to complete chemotherapy due to systemic ‘off target’ side effects.
• 1 / 3rd of cancer survivors suffer long-term side effects as a direct result of exposure to chemotherapy, including premature death.
• Most cancers rapidly develop resistance to current chemotherapies.
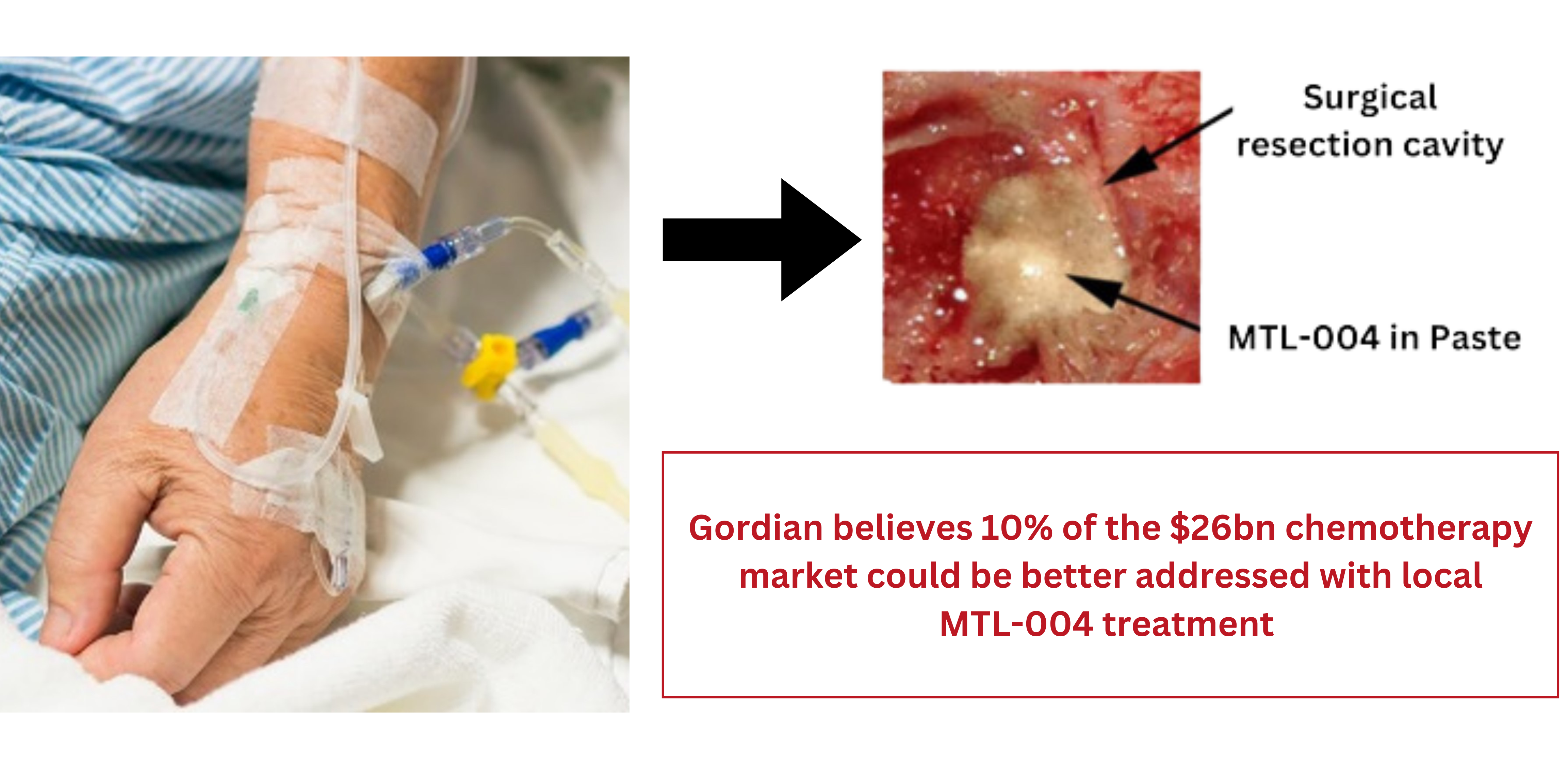
MTL-004 has Clear Advantages
- MTL-004 acts rapidly on contact with cells, initially deactivated with plasma (minimising systemic effects), then binding to cellular DNA in the target tissues.
- MTL-004 forms an exceptionally high frequency of covalent bonds (C8-O6) between DNA molecules (inter-strand crosslinks).
- Due to very limited repair of these inter-strand cross-links, they remain in-situ for an extended period (~25 days) or until cells attempt to divide, at which time novel apoptotic pathways are induced reliably leading to cell death, and the breakdown of MTL-004 itself.
- MTL-004 binds to DNA irrespective of genetic heterogeneity or disease stage, providing exceptionally broad and long-lasting control.
- There are no general toxicities observed in normal or cancer-bearing animals at the planned dose range for MTL-004.
MTL-004 leads to highly reliable cell death with broad potential for the localized treatment of solid tumours

Programme to Clinic – Readiness
December 1988: The Pro-drug of MTL-004 (CB 1954) was discovered at the ICR and trialled in man in combination with gene and enzyme therapy.
December 1992: MTL-004 was identified as the active component.
December 2006: MTL-004 Patents granted.

February 2021: The EMA provide guidance on non-clinical and CMC requirements under ICH-S9 for MTL-004 to be considered for clinical evaluation in Glioblastoma, Small cell lung cancer and invasive bladder
cancer.

June 2024: MHRA confirm acceptance of non-clinical and CMC data package subject to sterility and starting dose determination for a GBM study in man.

The Brain Tumour Charity are providing guidance on a pivotal post resection study in GBM to support an orphan drug application comparing tumour recurrence and overall survival of ~40 patients presenting with primary glioblastoma, receiving standard of care with or without initial treatment of the post-resection site with MTL-004 paste.
Gordian have focused on GBM as the
‘Lead Indication’ for MTL-004
Temozolomide
Orally administered Temozolomide is one of very few drugs licensed for treating brain cancers but has only limited efficacy, possibly extending survival by no more than a few months at the price of side effects including nausea, convulsions and lymphopenia.
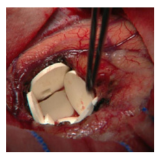
Carmustine
Wafers containing Carmustine, inserted at the resection site provide some protection against cancer recurrence, but tissue contact is variable, the drug is noxious / short-lived and results are variable/limited.
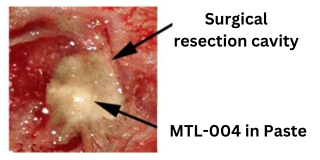
MTL – 004
The delivery of MTL-004 in a bioresorbable paste at the resection site provides excellent contact with the tumour margins ensuring extended tumour control and much improved survival in animals.
Gordian have demonstrated Safety, Toxicology & Tolerability at Therapeutic Doses
ADME studies
Confirm no hepatotoxicity detected in extensive range of assays.
hERG studies
Confirm no ion channel perturbation.
I.V. Dosing in Healthy Rats
Following repeated intravenous administration to rats during a 2 week study, the MTD (Maximum Tolerated Dose) of MTL-004 is considered to be 5 mg/kg/day, with a dose level of 1 mg/kg/day considered suitable for a study of longer duration.
4 week i.v. studies with repeat dosing at 1 mg/Kg showed no treatment-related toxicological effects in any organs or at the site of administration.
Post-resection treatment of Tumour Bearing Rats
Following resection of orthotopic tumours in rats intracranial dose escalation studies using 0.1-2.5 mg/Kg MTL-004 in the paste formulation revealed no differences in body weight or other physiological parameters between treated and control animals.
Contact Us
For enquiries about our products
